
Advanced examples
Lluís Revilla
lluis.revilla@gmail.com2026 May 18
Source:vignettes/advanced.Rmd
advanced.RmdAbstract
This vignette assumes you are familiar with set operations from the basic vignette.
Initial setup
To show compatibility with tidy workflows we will use magrittr pipe operator and the dplyr verbs.
Human gene ontology
We will explore the genes with assigned gene ontology terms. These terms describe what is the process and role of the genes. The links are annotated with different evidence codes to indicate how such annotation is supported.
# We load some libraries
library("org.Hs.eg.db", quietly = TRUE)
library("GO.db", quietly = TRUE)
library("ggplot2", quietly = TRUE)
# Prepare the data
h2GO_TS <- tidySet(org.Hs.egGO)
h2GO <- as.data.frame(org.Hs.egGO)We can now explore if there are differences in evidence usage for each ontology in gene ontology:
library("forcats", include.only = "fct_reorder2", quietly = TRUE)
h2GO %>%
group_by(Evidence, Ontology) %>%
count(name = "Freq") %>%
ungroup() %>%
mutate(Evidence = fct_reorder2(Evidence, Ontology, -Freq),
Ontology = case_match(Ontology,
"CC" ~ "Cellular Component",
"MF" ~ "Molecular Function",
"BP" ~ "Biological Process",
.default = NA)) %>%
ggplot() +
geom_col(aes(Evidence, Freq)) +
facet_grid(~Ontology) +
theme_minimal() +
coord_flip() +
labs(x = element_blank(), y = element_blank(),
title = "Evidence codes for each ontology")
#> Warning: There was 1 warning in `mutate()`.
#> ℹ In argument: `Ontology = case_match(...)`.
#> Caused by warning:
#> ! `case_match()` was deprecated in dplyr 1.2.0.
#> ℹ Please use `recode_values()` instead.
#> Warning: `label` cannot be a <ggplot2::element_blank> object.
#> `label` cannot be a <ggplot2::element_blank> object.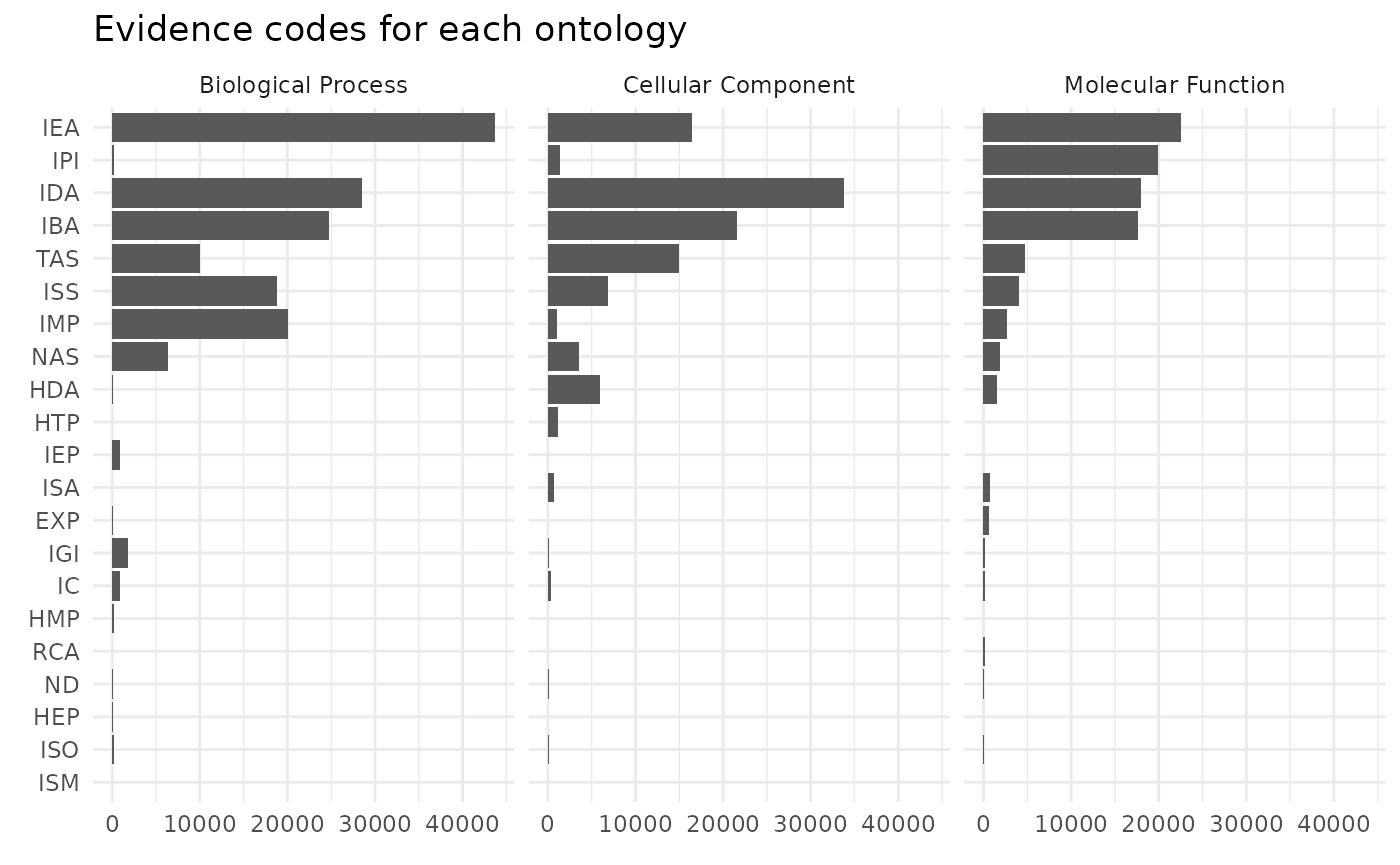
We can see that biological process are more likely to be defined by IMP evidence code that means inferred from mutant phenotype. While inferred from physical interaction (IPI) is almost exclusively used to assign molecular functions.
This graph doesn’t consider that some relationships are better annotated than other:
h2GO_TS %>%
relations() %>%
group_by(elements, sets) %>%
count(sort = TRUE, name = "Annotations") %>%
ungroup() %>%
count(Annotations, sort = TRUE) %>%
ggplot() +
geom_col(aes(Annotations, n)) +
theme_minimal() +
labs(x = "Evidence codes", y = "Annotations",
title = "Evidence codes for each annotation",
subtitle = "in human") +
scale_x_continuous(breaks = 1:7)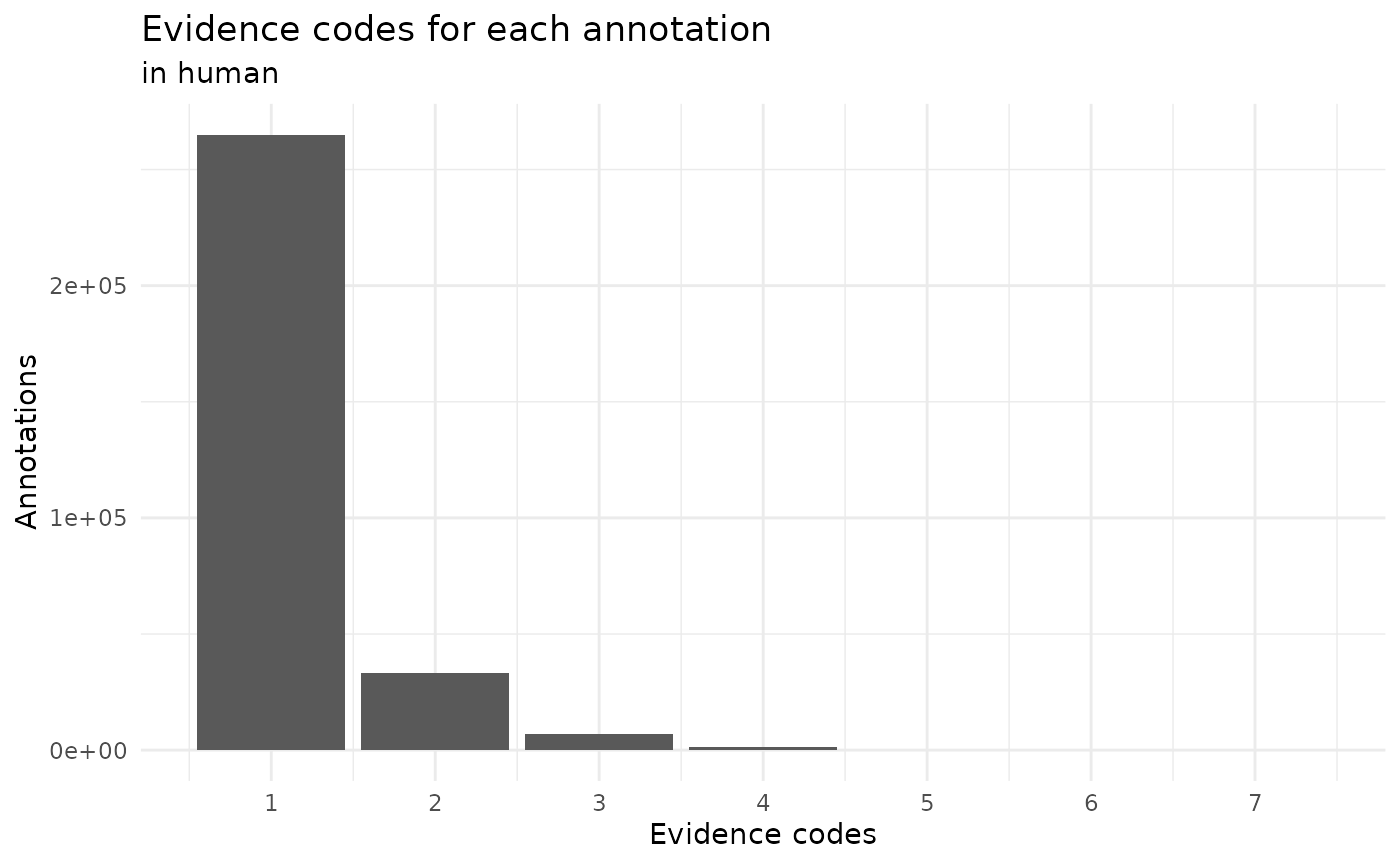
We can see that mostly all the annotations are done with a single evidence code. So far we have explored the code that it is related to a gene but how many genes don’t have any annotation?
# Add all the genes and GO terms
h2GO_TS <- add_elements(h2GO_TS, keys(org.Hs.eg.db)) %>%
add_sets(grep("^GO:", keys(GO.db), value = TRUE))
sizes_element <- element_size(h2GO_TS) %>%
arrange(desc(size))
sum(sizes_element$size == 0)
#> [1] 172971
sum(sizes_element$size != 0)
#> [1] 20826
sizes_set <- set_size(h2GO_TS) %>%
arrange(desc(size))
sum(sizes_set$size == 0)
#> [1] 20837
sum(sizes_set$size != 0)
#> [1] 17902So we can see that both there are more genes without annotation and more gene ontology terms without a (direct) gene annotated.
sizes_element %>%
filter(size != 0) %>%
ggplot() +
geom_histogram(aes(size), binwidth = 1) +
theme_minimal() +
labs(x = "# sets per element", y = "Count")
sizes_set %>%
filter(size != 0) %>%
ggplot() +
geom_histogram(aes(size), binwidth = 1) +
theme_minimal() +
labs(x = "# elements per set", y = "Count")
As you can see on the second plot we have very large values but that are on associated on many genes:
head(sizes_set, 10)
#> sets size probability Ontology
#> 1 GO:0005515 13662 1 MF
#> 2 GO:0005737 6561 1 CC
#> 3 GO:0005634 6531 1 CC
#> 4 GO:0016020 5581 1 CC
#> 5 GO:0005886 5472 1 CC
#> 6 GO:0005829 5087 1 CC
#> 7 GO:0046872 4053 1 MF
#> 8 GO:0005654 3641 1 CC
#> 9 GO:0005576 2458 1 CC
#> 10 GO:0003677 2296 1 MFUsing fuzzy values
This could radically change if we used fuzzy values. We could assign a fuzzy value to each evidence code given the lowest fuzzy value for the IEA (Inferred from Electronic Annotation) evidence. The highest values would be for evidence codes coming from experiments or alike.
nr <- h2GO_TS %>%
relations() %>%
dplyr::select(sets, Evidence) %>%
distinct() %>%
mutate(fuzzy = case_match(Evidence,
"EXP" ~ 0.9,
"IDA" ~ 0.8,
"IPI" ~ 0.8,
"IMP" ~ 0.75,
"IGI" ~ 0.7,
"IEP" ~ 0.65,
"HEP" ~ 0.6,
"HDA" ~ 0.6,
"HMP" ~ 0.5,
"IBA" ~ 0.45,
"ISS" ~ 0.4,
"ISO" ~ 0.32,
"ISA" ~ 0.32,
"ISM" ~ 0.3,
"RCA" ~ 0.2,
"TAS" ~ 0.15,
"NAS" ~ 0.1,
"IC" ~ 0.02,
"ND" ~ 0.02,
"IEA" ~ 0.01,
.default = 0.01)) %>%
dplyr::select(sets = "sets", elements = "Evidence", fuzzy = fuzzy)We have several evidence codes for the same ontology, this would result on different fuzzy values for each relation. Instead, we extract this and add them as new sets and elements and add an extra column to classify what are those elements:
ts <- h2GO_TS %>%
relations() %>%
dplyr::select(-Evidence) %>%
rbind(nr) %>%
tidySet() %>%
mutate_element(Type = ifelse(grepl("^[0-9]+$", elements), "gene", "evidence"))Now we can see which gene ontologies are more supported by the evidence:
ts %>%
dplyr::filter(Type != "Gene") %>%
cardinality() %>%
arrange(desc(cardinality)) %>%
head()
#> sets cardinality
#> 1 GO:0005515 13664.90
#> 2 GO:0005737 6567.00
#> 3 GO:0005634 6537.00
#> 4 GO:0016020 5585.28
#> 5 GO:0005886 5477.28
#> 6 GO:0005829 5092.68Surprisingly the most supported terms are protein binding, nucleus and cytosol. I would expect them to be the top three terms for cellular component, biological function and molecular function.
Calculating set sizes would be interesting but it requires computing a big number of combinations that make it last long and require many memory available.
ts %>%
filter(sets %in% c("GO:0008152", "GO:0003674", "GO:0005575"),
Type != "gene") %>%
set_size()
#> sets size probability
#> 1 GO:0003674 0 0.98
#> 2 GO:0003674 1 0.02
#> 3 GO:0005575 0 0.98
#> 4 GO:0005575 1 0.02Unexpectedly there is few evidence for the main terms:
go_terms <- c("GO:0008152", "GO:0003674", "GO:0005575")
ts %>%
filter(sets %in% go_terms & Type != "gene")
#> elements sets fuzzy Type
#> 1 ND GO:0005575 0.02 evidence
#> 2 ND GO:0003674 0.02 evidenceIn fact those terms are arbitrarily decided or inferred from electronic analysis.
Human pathways
Now we will repeat the same analysis with pathways:
# We load some libraries
library("reactome.db")
# Prepare the data (is easier, there isn't any ontoogy or evidence column)
h2p <- as.data.frame(reactomeEXTID2PATHID)
colnames(h2p) <- c("sets", "elements")
# Filter only for human pathways
h2p <- h2p[grepl("^R-HSA-", h2p$sets), ]
# There are duplicate relations with different evidence codes!!:
summary(duplicated(h2p[, c("elements", "sets")]))
#> Mode FALSE TRUE
#> logical 139784 16545
h2p <- unique(h2p)
# Create a TidySet and
h2p_TS <- tidySet(h2p) %>%
# Add all the genes
add_elements(keys(org.Hs.eg.db))Now that we have everything ready we can start measuring some things…
sizes_element <- element_size(h2p_TS) %>%
arrange(desc(size))
sum(sizes_element$size == 0)
#> [1] 182510
sum(sizes_element$size != 0)
#> [1] 11686
sizes_set <- set_size(h2p_TS) %>%
arrange(desc(size))We can see there are more genes without pathways than genes with pathways.
sizes_element %>%
filter(size != 0) %>%
ggplot() +
geom_histogram(aes(size), binwidth = 1) +
scale_y_log10() +
theme_minimal() +
labs(x = "# sets per element", y = "Count")
#> Warning in scale_y_log10(): log-10 transformation introduced
#> infinite values.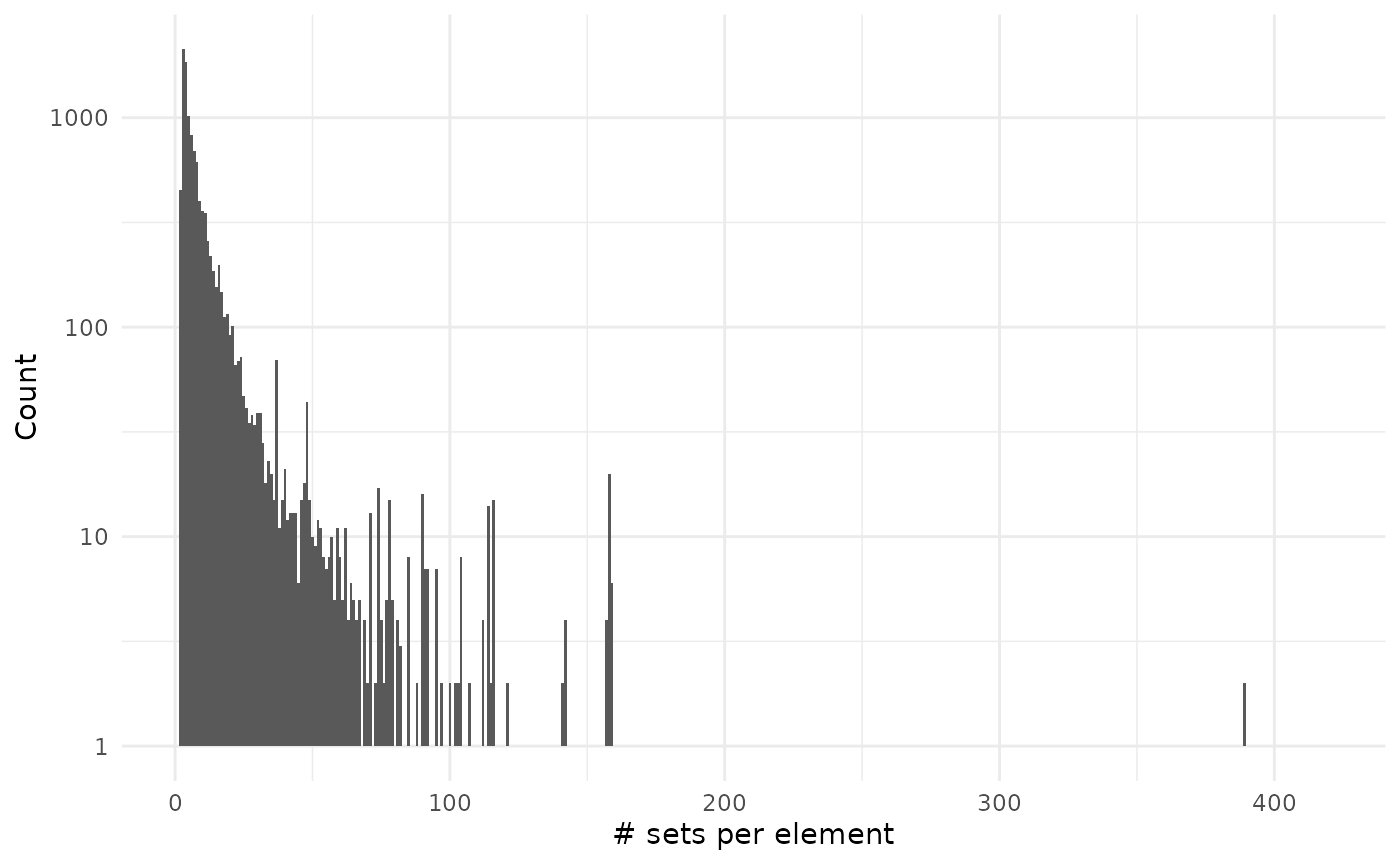
sizes_set %>%
ggplot() +
geom_histogram(aes(size), binwidth = 1) +
scale_y_log10() +
theme_minimal() +
labs(x = "# elements per set", y = "Count")
#> Warning in scale_y_log10(): log-10 transformation introduced
#> infinite values.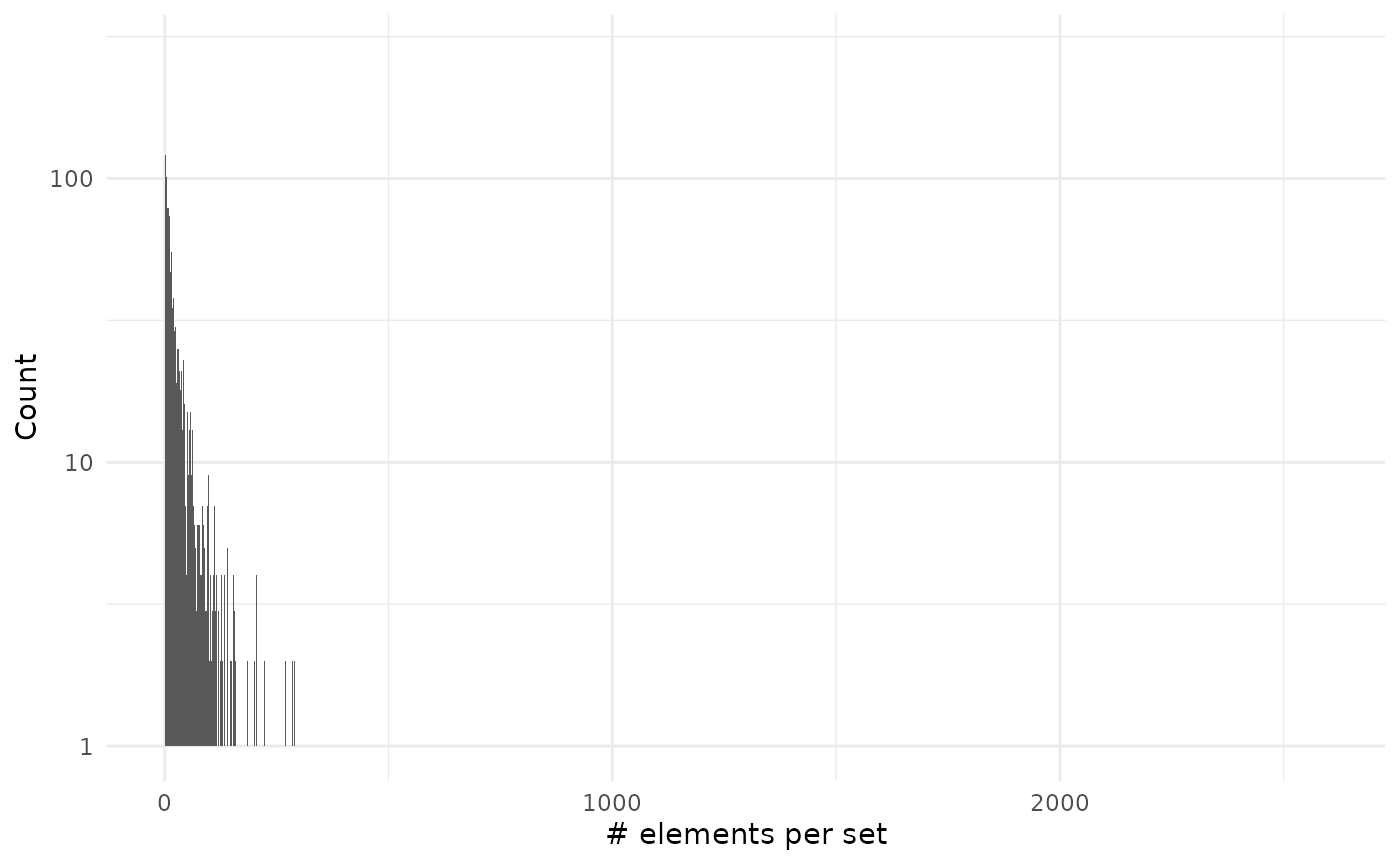
As you can see on the second plot we have very large values but that are on associated on many genes:
head(sizes_set, 10)
#> sets size probability
#> 1 R-HSA-162582 2609 1
#> 2 R-HSA-1643685 2446 1
#> 3 R-HSA-1430728 2186 1
#> 4 R-HSA-168256 2176 1
#> 5 R-HSA-392499 2128 1
#> 6 R-HSA-5663205 1633 1
#> 7 R-HSA-1266738 1534 1
#> 8 R-HSA-74160 1520 1
#> 9 R-HSA-597592 1500 1
#> 10 R-HSA-73857 1322 1Session info
#> R version 4.6.0 (2026-04-24)
#> Platform: x86_64-pc-linux-gnu
#> Running under: Ubuntu 24.04.4 LTS
#>
#> Matrix products: default
#> BLAS: /usr/lib/x86_64-linux-gnu/openblas-pthread/libblas.so.3
#> LAPACK: /usr/lib/x86_64-linux-gnu/openblas-pthread/libopenblasp-r0.3.26.so; LAPACK version 3.12.0
#>
#> locale:
#> [1] LC_CTYPE=en_US.UTF-8 LC_NUMERIC=C
#> [3] LC_TIME=en_US.UTF-8 LC_COLLATE=en_US.UTF-8
#> [5] LC_MONETARY=en_US.UTF-8 LC_MESSAGES=en_US.UTF-8
#> [7] LC_PAPER=en_US.UTF-8 LC_NAME=C
#> [9] LC_ADDRESS=C LC_TELEPHONE=C
#> [11] LC_MEASUREMENT=en_US.UTF-8 LC_IDENTIFICATION=C
#>
#> time zone: Etc/UTC
#> tzcode source: system (glibc)
#>
#> attached base packages:
#> [1] stats4 stats graphics grDevices utils datasets methods
#> [8] base
#>
#> other attached packages:
#> [1] reactome.db_1.96.0 forcats_1.0.1 ggplot2_4.0.3
#> [4] GO.db_3.23.1 org.Hs.eg.db_3.23.1 AnnotationDbi_1.75.0
#> [7] IRanges_2.47.1 S4Vectors_0.51.2 Biobase_2.73.1
#> [10] BiocGenerics_0.59.2 generics_0.1.4 dplyr_1.2.1
#> [13] BaseSet_1.0.0.9001
#>
#> loaded via a namespace (and not attached):
#> [1] KEGGREST_1.53.0 gtable_0.3.6 xfun_0.57 bslib_0.11.0
#> [5] vctrs_0.7.3 tools_4.6.0 tibble_3.3.1 RSQLite_3.52.0
#> [9] blob_1.3.0 pkgconfig_2.0.3 RColorBrewer_1.1-3 S7_0.2.2
#> [13] desc_1.4.3 graph_1.91.0 lifecycle_1.0.5 compiler_4.6.0
#> [17] farver_2.1.2 textshaping_1.0.5 Biostrings_2.81.1 Seqinfo_1.3.0
#> [21] htmltools_0.5.9 sass_0.4.10 yaml_2.3.12 pkgdown_2.2.0
#> [25] pillar_1.11.1 crayon_1.5.3 jquerylib_0.1.4 cachem_1.1.0
#> [29] tidyselect_1.2.1 digest_0.6.39 labeling_0.4.3 fastmap_1.2.0
#> [33] grid_4.6.0 cli_3.6.6 magrittr_2.0.5 XML_3.99-0.23
#> [37] GSEABase_1.75.0 withr_3.0.2 scales_1.4.0 bit64_4.8.0
#> [41] rmarkdown_2.31 XVector_0.53.0 httr_1.4.8 bit_4.6.0
#> [45] otel_0.2.0 ragg_1.5.2 png_0.1-9 memoise_2.0.1
#> [49] evaluate_1.0.5 knitr_1.51 rlang_1.2.0 xtable_1.8-8
#> [53] glue_1.8.1 DBI_1.3.0 annotate_1.91.0 jsonlite_2.0.0
#> [57] R6_2.6.1 systemfonts_1.3.2 fs_2.1.0